 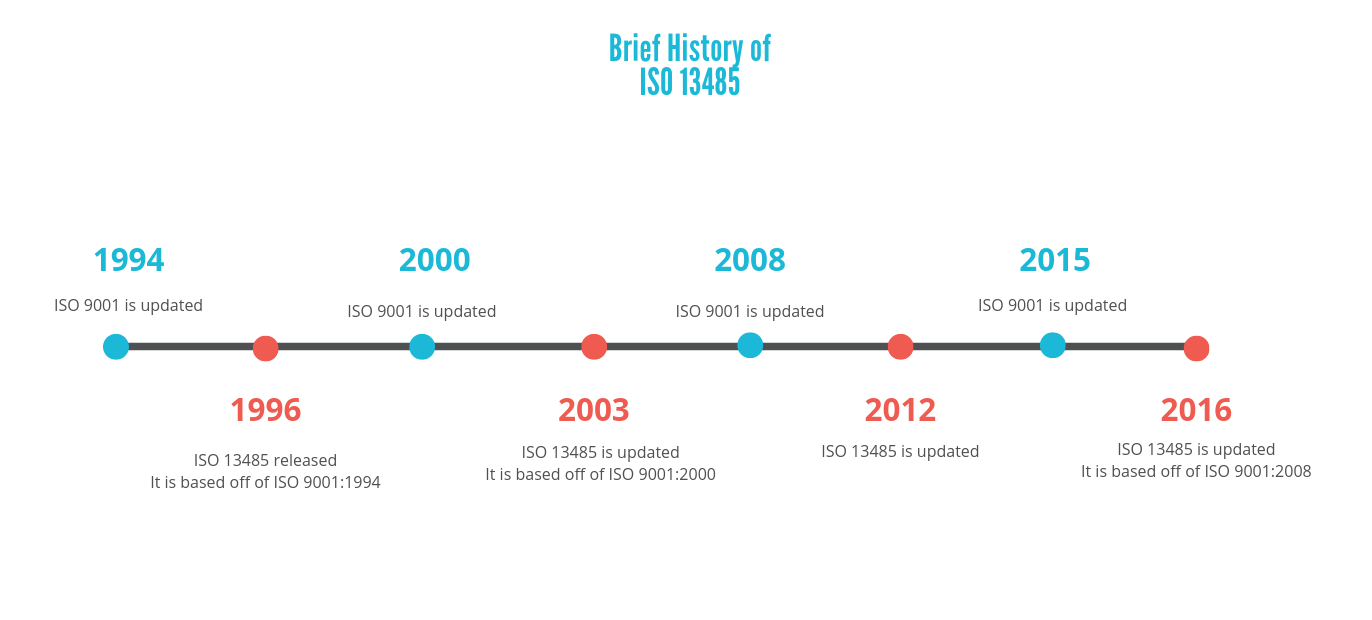 
In order to understand the utilization of each standard today, it is important to understand how each of these standards was developed. Commercialization of medical devices in the United States requires compliance with Good Manufacturing Practices (GMPs) defined in 21 CFR 820, which are federal regulations governed by the U.S. The internationally recognized standard for quality management systems is ISO 9001, specifically ISO 13485 for Medical Devices. There are standards in place to achieve each of these aims within a company. The quality management system of a medical device manufacturer is important for several reasons, but today we are going to focus on its aims of customer satisfaction and device safety and effectiveness.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
